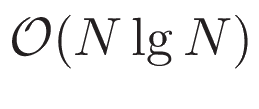
A number of proposals emerged to explain unexpectedly long coherence. Single molecule spectroscopy now shows the quantum characteristics of photosynthesis without the interference of static disorder, and some studies use this method to assign reported signatures of electronic quantum coherence to nuclear dynamics occurring in chromophores. However, critical follow-up studies question the interpretation of these results. These studies suggest that, through evolution, nature has developed a way of protecting quantum coherence to enhance the efficiency of photosynthesis. In that same year, experiments conducted on photosynthetic cryptophyte algae using two-dimensional photon echo spectroscopy yielded further confirmation for long-term quantum coherence. Another theoretical study from 2010 provided evidence that quantum coherence lives as long as 300 femtoseconds at biologically relevant temperatures (4 ☌ or 277 K). A study published in 2007 claimed the identification of electronic quantum coherence at −196 ☌ (77 K). Scientists have recently looked for experimental evidence of this proposed energy transfer mechanism. Instead, as early as 1938, scientists theorized that quantum coherence was the mechanism for excitation energy transfer. FT electron spectroscopy studies of electron absorption and transfer show an efficiency of above 99%, which cannot be explained by classical mechanical models like the diffusion model. Various structures, such as the FMO complex in green sulfur bacteria, are responsible for transferring energy from antennae to a reaction site. The energy collected in reaction sites must be transferred quickly before it is lost to fluorescence or thermal vibrational motion. Photosynthesis creates Frenkel excitons, which provide a separation of charge that cells convert into usable chemical energy. For example, bacteria use ring-like antennae, while plants use chlorophyll pigments to absorb photons. Organisms that undergo photosynthesis absorb light energy through the process of electron excitation in antennae. The excitation then transfers through various proteins in the FMO complex to the reaction center to further photosynthesis. Applications Photosynthesis ĭiagram of FMO complex. 
In 1979, the Soviet and Ukrainian physicist Alexander Davydov published the first textbook on quantum biology entitled Biology and Quantum Mechanics. In his paper, he stated that there is a new field of study called "quantum biology". In 1963, Per-Olov Löwdin published proton tunneling as another mechanism for DNA mutation. Other pioneers Niels Bohr, Pascual Jordan, and Max Delbrück argued that the quantum idea of complementarity was fundamental to the life sciences. 
He further suggested that mutations are introduced by "quantum leaps". Schrödinger introduced the idea of an " aperiodic crystal" that contained genetic information in its configuration of covalent chemical bonds. Erwin Schrödinger's 1944 book What Is Life? discussed applications of quantum mechanics in biology. Early pioneers of quantum physics saw applications of quantum mechanics in biological problems. It has been suggested that quantum biology might play a critical role in the future of the medical world. Though the field has only recently received an influx of attention, it has been conceptualized by physicists throughout the 20th century. Quantum biology is an emerging field, in the sense that most current research is theoretical and subject to questions that require further experimentation. Quantum biology is concerned with the influence of non-trivial quantum phenomena, which can be explained by reducing the biological process to fundamental physics, although these effects are difficult to study and can be speculative. Quantum biology may use computations to model biological interactions in light of quantum mechanical effects. Such processes involve chemical reactions, light absorption, formation of excited electronic states, transfer of excitation energy, and the transfer of electrons and protons ( hydrogen ions) in chemical processes, such as photosynthesis, olfaction and cellular respiration. Many biological processes involve the conversion of energy into forms that are usable for chemical transformations, and are quantum mechanical in nature. An understanding of fundamental quantum interactions is important because they determine the properties of the next level of organization in biological systems. 
Quantum biology is the study of applications of quantum mechanics and theoretical chemistry to aspects of biology that cannot be accurately described by the classical laws of physics. Application of quantum mechanics and theoretical chemistry to biological objects and problems
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
